 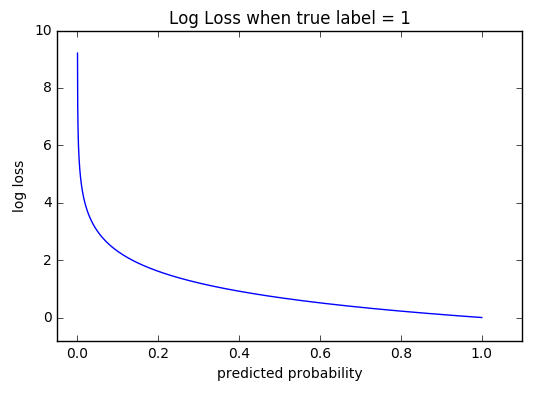
We first show by straightforward calculation that the phase volume Gamma (E,V,N). The fact that the absolute value of specific entropy is unknown is not a problem because it is the change in specific entropy (Δs) and not the absolute value that is important in practical problems. Derivation of the entropy formula for the microcanonical ensemble. For example, the specific entropy of water or steam is given using the reference that the specific entropy of water is zero at 32☏. Also, like enthalpy, the entropy of a substance is given with respect to some reference value. Like enthalpy, entropy cannot be measured directly. Entropy is represented by the letter S and can be defined as ΔS in the following relationships. Entropy is sometimes referred to as a measure of the inability to do work for a given heat transferred. Because entropy tells so much about the usefulness of an amount of heat transferred in performing work, the steam tables include values of specific entropy (s = S/m) as part of the information tabulated. Entropy quantifies the energy of a substance that is no longer available to perform useful work. Any UV finite theory of quantum gravity should reduce at low energy to General Relativity. Because entropy is a property, changes in it can be determined by knowing the initial and final conditions of a substance. The Hawking formula for the entropy receives corrections as soon as quantum effects are taken into account. It is the configuration corresponding to the maximum of entropy at equilibrium. Boltzmann's entropy describes the system when all the accessible microstates are equally likely. 
> Thermodynamics Directory | Heat Transfer DirectoryĮntropy Definition - Thermodynamic PropertiesĮntropy (S) is a property of a substance, as are pressure, temperature, volume, and enthalpy. Since is a natural number(1,2,3.), entropy is either zero or positive (ln 1 0, ln 0). R., 2010, Advanced Heat and Mass Transfer, Global Digital Press, Columbia, MO.Entropy Definition and Equation Thermodynamics (-log1 0) Note that thermodynamic entropy and the entropy in information theory both capture increasing randomness. show such entropy formula and derive some applications of the new entropy formula. The second law of thermodynamics requires that each of these entropy generations be greater than or equal to zero.įaghri, A., and Zhang, Y., 2006, Transport Phenomena in Multiphase Systems, Burlington, MA.įaghri, A., Zhang, Y., and Howell, J. The entropy generation for a control volume including Π phases consists of entropy generation in each phase, plus that in the interfaces. If the control volume includes Π phases, the second law of thermodynamics must be obtained by integrating over the two phases separately (Faghri and Zhang, 2006), Consider a data set having a total number of N classes, then the entropy (E) can be determined with the formula below. (3), one obtains the integral form of the second law of thermodynamics for single phase systems: The last term represents entropy generation, which should always be greater than or equal to zero, i.e.,Ĭombining eqs. (2), the first term represents the change of entropy due to heat transfer across the boundary of the control volume, and the second term represents the change of entropy due to internal heat generation in the control volume. The change of entropy in a closed system results from heat transfer and/or entropy generation: 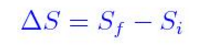
The entropy change for a system with fixed-mass and only one phase can be obtained by setting Φ = S,φ = s in eq. The second law of thermodynamics requires that the entropy generation in a closed system (fixed-mass) must be greater than or equal to zero. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
